- Blog
- Time for writing
- Minimized windows on top of taskbar
- Spirit animal by birthday in telugu
- Burnout 3 takedown crash mode
- The conscious reader 12th edition
- Hitman go 1-14 no kill
- Transfer ost to pst
- Yu gi oh 7 trials to glory gameshark codes
- Tomb raider lara croft nude game
- Neofinder thumbnails not showing
- Satta matka king com
- Hp officejet pro 8720 scan to email setup
- Small crane lift
- American masters episodes
- French cafe music instrumental
- Welch allyn spot vital signs lxi 45oto with accessories
- What is electron affinity
- Blog
- Time for writing
- Minimized windows on top of taskbar
- Spirit animal by birthday in telugu
- Burnout 3 takedown crash mode
- The conscious reader 12th edition
- Hitman go 1-14 no kill
- Transfer ost to pst
- Yu gi oh 7 trials to glory gameshark codes
- Tomb raider lara croft nude game
- Neofinder thumbnails not showing
- Satta matka king com
- Hp officejet pro 8720 scan to email setup
- Small crane lift
- American masters episodes
- French cafe music instrumental
- Welch allyn spot vital signs lxi 45oto with accessories
- What is electron affinity

(More negative energy = greater affinity) Element Obviously, the halogens, which are one electron away from a noble gas electron configuration, have high affinities for electrons:
#What is electron affinity free#
Since a half-filled "p" subshell is more stable, carbon has a greater affinity for an electron than nitrogen. The electron affinity is the energy required to raise the electron from the the bottom of the conduction band to the vacuum free electron level. Answer (1 of 2): Electron affinity means the energy released when 1 mole of neutral atoms of an element each receives one electron (thus 1 mole electron in total) in gaseous state and become 1 mole of negative ion. Electron affinity definition, the quantitative measure, usually given in electron-volts, of the tendency of an atom or molecule to capture an electron and to form a negative ion. This occurs because of the same subshell rule that governs ionization energies. The change is small and there are many exceptions.Įlectron affinity decreases or increases across a period depending on electronic configuration. In other words, EA denotes the probability of a neutral atom acquiring an electron. (An unbound electron has an energy of zero.)Īs with ionization energy, there are two rules that govern the periodic trends of electron affinities:Įlectron affinity becomes less negative down a group.Īs the principal quantum number increases, the size of the orbital increases and the affinity for the electron is less. Electron affinity is defined as the change in energy (kJ/mole) of a neutral atom (in the gaseous phase) when one electron is added to it to form a negative ion (EA). Lithium salt of carboxylic acid is not used in Kolbe's electrolysis where as Sodium salt of carboxylic acid is used. A more negative electron affinity corresponds to a greater attraction for an electron.
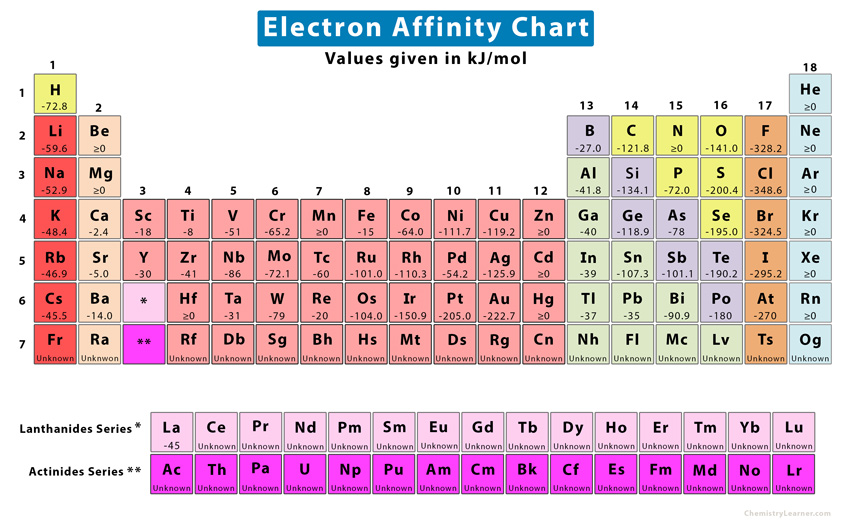
This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. This is the reverse of the electron affinity of the. For example, when a fluorine atom in the gaseous state gains an electron to form F(g), the associated energy change is -328 kJ/mol. Notice the sign on the energy is negative. Double affinity is when by means of four bodies, two decompositions and two new combinations are. Electron affinity is the energy change that results from adding an electron to a gaseous atom.
